The LogiPharma conference held in spring every year is the premier event focused on all things supply chain and distribution in the pharmaceutical industry. This year it was in Basel. The first thing I have learned over the past winter has been the impact of the Polar vortex and the fact that despite Basel being considerably further north than Toronto, spring can arrive a lot earlier, thanks also to the North Atlantic Drift. But I am getting distracted.
Lesson 1: Control Towers are becoming mainstream
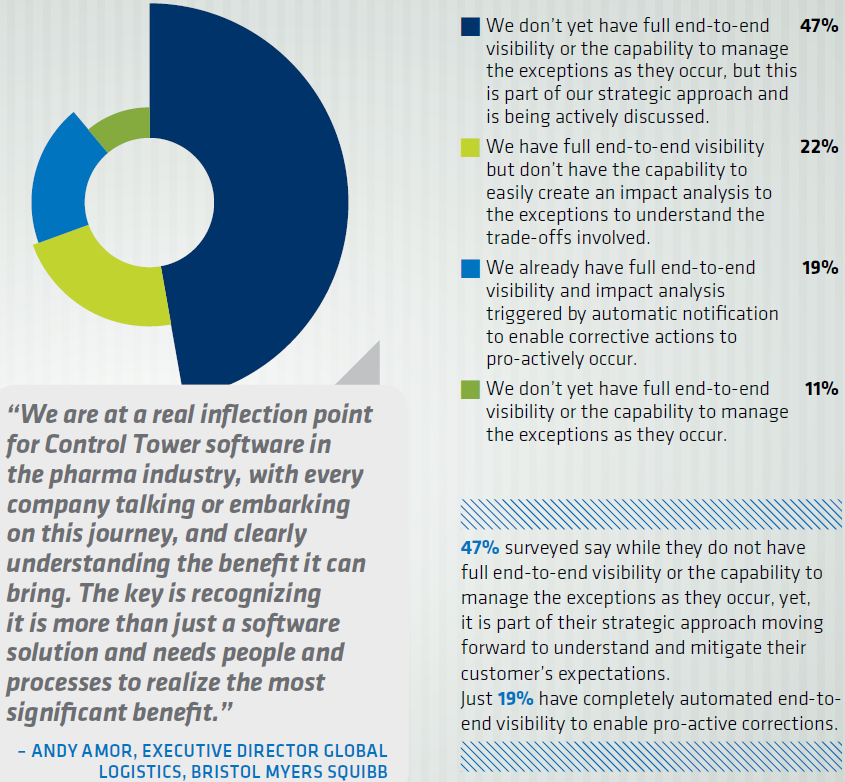
In the case of LogiPharma they had a really interesting survey about the state of pharmaceutical supply chain capabilities and needs. A core theme of end-to-end planning and visibility that jumped out of the preconference survey continued throughout the conference. As per the diagram extracted from the survey, the term used by many people to encapsulate this idea of end-to-end planning and visibility is ‘control tower’.
What is interesting is how this theme is so consistent with Gartner’s most recent Magic Quadrant for Supply Chain Planning Systems of Record. In the report Gartner defines a supply chain planning (SCP) System of Record (SOR) as:
An SCP SOR is a planning platform that enables a company to create, manage, link, align, collaborate and share its planning data across a supply chain — from demand plan creation through the supply-side response, and from detailed operational planning through tactical-level planning. [i]
However, where a ‘control tower’ comes into play is when Gartner goes on to state that:
Increasingly the deployment of a SCP SOR will have to be in the context of multi-enterprise supply chains and the convergence of planning and execution to facilitate more responsive, agile planning – especially in the short-term horizon – across extended value chains. [ii]
In other words we are seeing the two ends of the supply chain – demand and supply – extend beyond the traditional span of control of the pharmaceutical manufacturers. On the demand side this is due to a number of factors, but the biggest drivers are expansion into global markets on the demand side and the relative explosion of outsourcing on the supply side. As the supply chain extends beyond the boundaries of the pharmaceutical manufacturers they have less and less visibility because typically they rely on their ERP solution, but as Jim Cafone, Vice President, Supply Network Services at Pfizer Pharmaceuticals said a few years ago at the Gartner Supply Chain Leaders conference:
ERP is fine if you only want to talk to yourself. But who can afford to do that in today’s world?
While Pfizer have deployed a logistics control tower through our partner GT Nexus, they have not yet matured to a Supply Chain Planning SOR as described by Gartner. Of course every increment in capability needs to be celebrated, and what Pfizer has done is a big leap forward, there is still some ways to go for them to be able to plan across multiple tiers, not just execute. Which brings me to the second thing I learned at LogiPharma…
Lesson 2: The definition of a Control Tower is evolving
The survey shows clearly that in general the idea of end-end planning has gained a lot of attention, but that the realization of this vision is some way off with 47% of companies saying that they have neither end-to-end visibility nor the ability to manage exceptions. A total of 80% (47% + 22% + 11%) don’t have the capability to create and impact analysis to exceptions. It is the remaining 20% (or 19% according to the survey results) that fascinate me. What exactly are they doing that allows them to claim that
We already have full end-to-end visibility and impact analysis triggered by automatic notification to enable corrective actions to pro-actively occur.
Here is a problem with all self-assessment surveys: The terms are never defined very well, if at all. In the statement above:
- What does end-to-end visibility mean? Does it mean from receipt of API (from a CMO) to shipment of finished product to an affiliate? Or does it include tracking of API production through the CMO and visibility into the affiliates inventories and sell-out demand?
- How long does it take to calculate the impact? Is it that a shipment will arrive later than expected? Or does this mean that you can also calculate the impact of the late shipment on revenue (broad) and customer service (specific)? And determine the specific people who should know about the initial cause and who are responsible for the impact?
- What constitutes corrective action occurring proactively? Does this mean that you can call up the logistics company and complain? Or does this mean that you can bring together a group of people in a virtual end-to-end environment to run rapid what-if analysis so that they can understand the business trade-offs they need to make across competing metrics?
Companies with very different performance and process maturity could have selected the same answer. I am pretty certain that few companies will have reached the level of maturity described by Gartner in the Planning SOR, definitely less than 20%. But I am encouraged by the fact that 80% of companies recognize the need to improve, because improvement starts with the recognition of the need to improve.
Lesson 3: It requires a certain type of person to get there
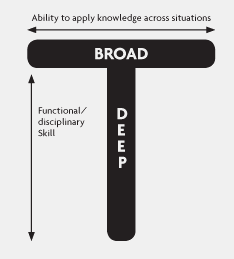
In my opinion the best presentation at LogiPharma was given by Hanns-Eberhard Erle of Merck Serono on “Elevating the supply chain function to meet the larger business needs”. This is at the core of every company’s need for end-to-end supply chain visibility and planning. This is the essence of what a mature supply chain planning Control Tower capability provides a company. The key to Hanns’ presentation was the observation that we need to develop T-shaped people. The idea of a T-shaped person , which is someone who has both a deep functional expertise and an understanding of how his/her role fits into the overall business , has been around for some time, but Hanns brought it to life in the context of a supply chain planning Control Tower. Hanns’ contention is that in the Life Sciences industries we tend to celebrate the specialist too much, the I-shaped person. His solution is to rotate people through the different supply chain functions, and by that he meant through manufacturing, quality, commercial, regulatory, etc., functions which people do not necessarily associate with supply chain, but which have a big impact on lead times, profitability, and customer service. I agree. What I observe is that because the Life Sciences industry is so heavily regulated the tendency is to keep people within a function to ensure that they have a deep understanding of the process, regulations, and technology. But this is counterproductive because we have done is create very soloed processes which are in fact cross-functional. Hann’s answer is to rotate people through the functions so that they get a much better understanding of the impact of decisions made in one function and multiple other functions. In a Harvard Business Review article titled “Introducing T-Shaped Managers: Knowledge Management’s Next Generation” by Morten T. Hansen and Bolko von Oetinger and dating from March 2001, state the following about becoming T-shaped:
It relies on a new kind of executive, one who breaks out of the traditional corporate hierarchy to share knowledge freely across the organization (the horizontal part of the “T”) while remaining fiercely committed to individual business unit performance (the vertical part). The successful T-shaped manager must learn to live with, and ultimately thrive within, the tension created by this dual responsibility. Although this tension is most acute for heads of business units, any T-shaped manager with operating unit obligations must wrestle with it.
I contend that it is only in the beginning, in early stages of maturity while the silos still dominate, that there is a tension in this role as described by HBR. A more mature organization, in which T-shaped mentality is the norm, will experience less tension because people will understand the business trade-offs and more time can be spent exploring the best cross-functional compromises and less time can be spent defending function specific metrics.
What’s Next?
In closing, at LogiPharma I presented on “Continuous S&OP – Breaking the Mold”. S&OP has been around for about 30 years and one of the original objectives was to bring the sales or commercial organization together with the manufacturing or supply side of the organization. I don’t know about you, but this sounds like the early stages of end-to-end supply chain planning. And I contend that it is, but that our idea about S&OP being a rigid monthly process has to change. Concepts such as a supply chain planning Control Tower provide the horizontal part of the T, but Gartner’s Supply Chain Planning SOR adds a third dimension by including the need to plan across different time horizons “…from detailed operational planning through tactical-level planning.” This is where I believe the next wave of innovation in supply chain planning will be.
Disclaimer: Gartner does not endorse any vendor, product or service depicted in its research publications, and does not advise technology users to select only those vendors with the highest ratings. Gartner research publications consist of the opinions of Gartner’s research organization and should not be construed as statements of fact. Gartner disclaims all warranties, expressed or implied, with respect to this research, including any warranties of merchantability or fitness for a particular purpose.
Additional Resources
- Supply chain control tower frequently asked questions




Leave a Reply